 The alkaline metals react with water at room temperature to produce hydroxides that are almost insoluble in water, along with hydrogen ions to form a basic solution. īeryllium (Be) does not react with water due to its small atomic size and relatively high ionization energy. Since all the alkaline metals have only two valence electrons, it takes little energy to make them give up those electrons to form cations (2+), resulting in high reactivity. 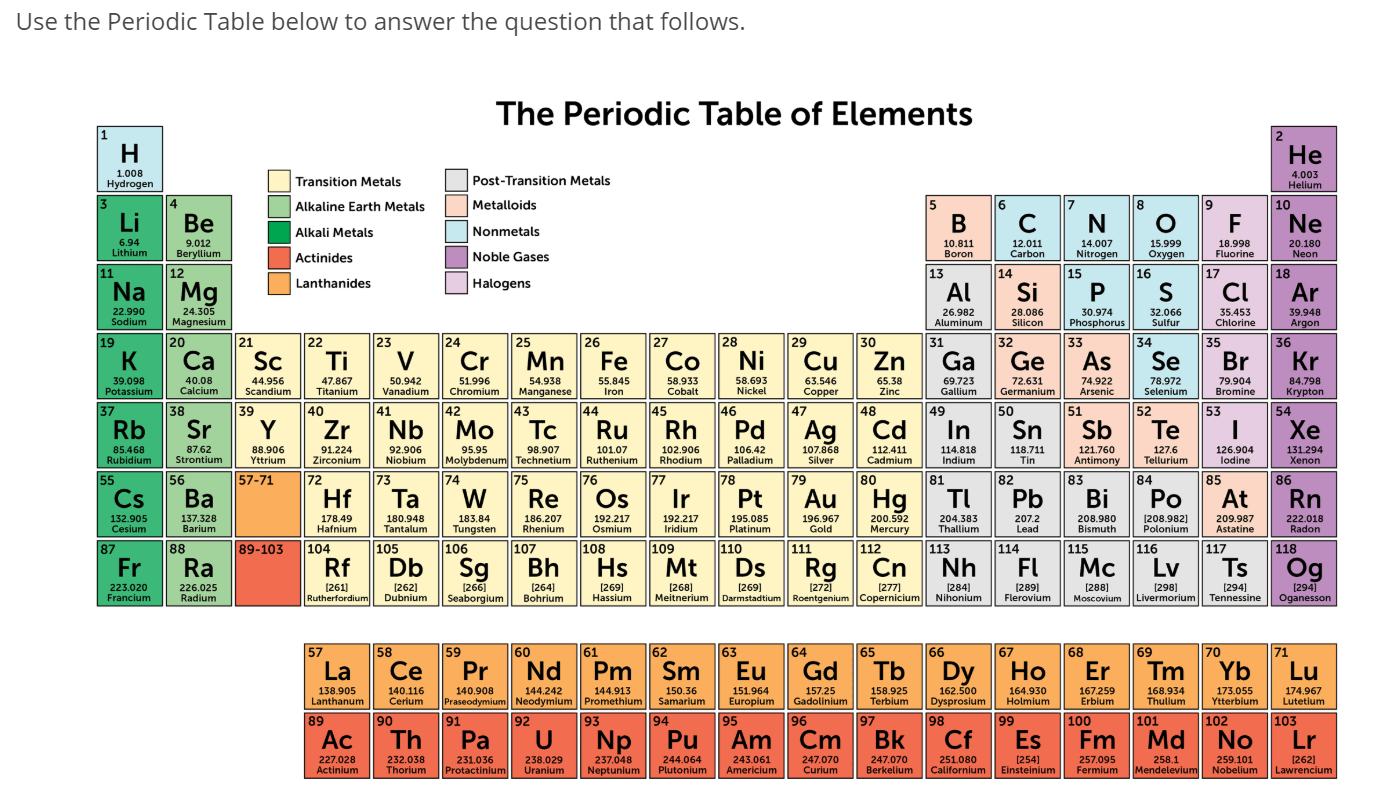
The lower the ionization energy, the more reactive an element is. The energy required to make an atom give up the electrons in its outermost shell (valence electrons) is the ionization energy for the element. Why are the Alkaline Earth Metals so Reactive
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
